Ideal Gas Assumptions - Kinetic Theory
5 (800) · $ 15.50 · In stock
When considering a gas as an ideal gas and applying the ideal gas law pV=nRT, we need to make 4 assumptions. (1) The volume of a molecule within the gas is n

Solved A. Using the assumptions of the kinetic theory of

Kinetic Theory Of Gases - Notes - LearnPick India
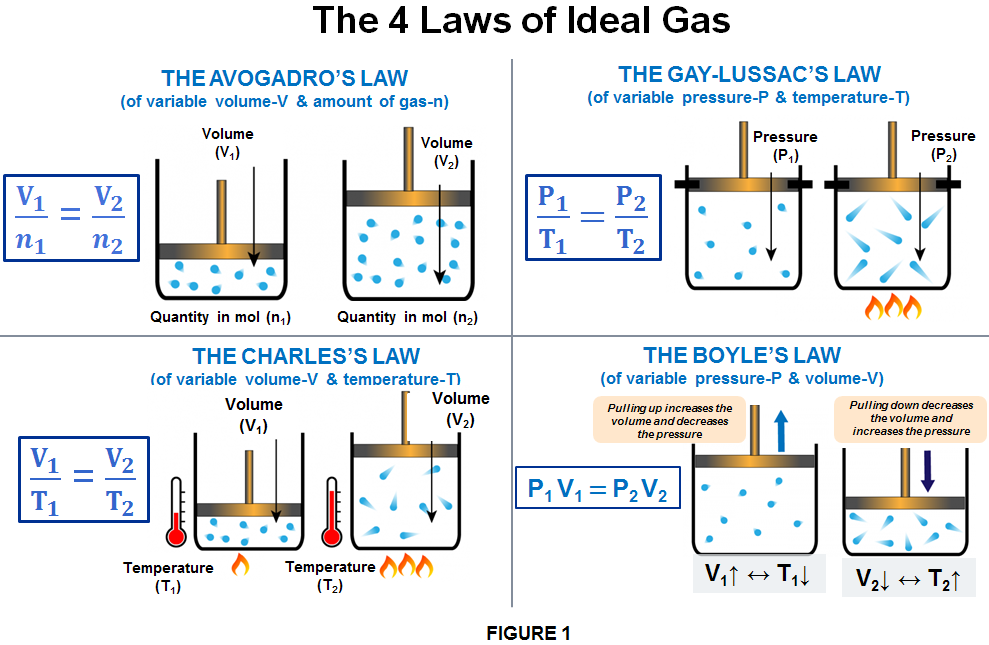
THE 3rd STATE OF MATTER – What is an Ideal Gas? – Computer Aided
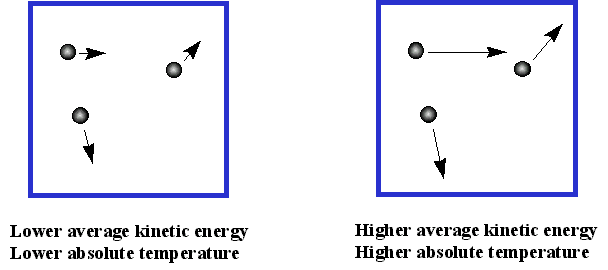
Basics of Kinetic Molecular Theory - Chemistry LibreTexts

1-4 Gases (Part 3)

What does point mass mean?

Kinetic Theory of Gases Formula, Assumptions & History - Physics
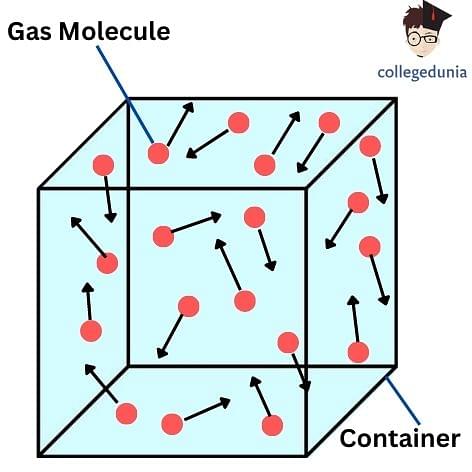
Important Kinetic Theory of Gas Formulas

quantum gases
Molecular Kinetic Theory Model Worksheets, Questions and Revision

Kinetic Molecular Theory Definition, Assumptions & Examples

01 Kinetic Theory of Gases Theory1

Kinetic Theory Class 11 Notes CBSE Physics Chapter 13 (PDF)

Ideal Gas Introduction











