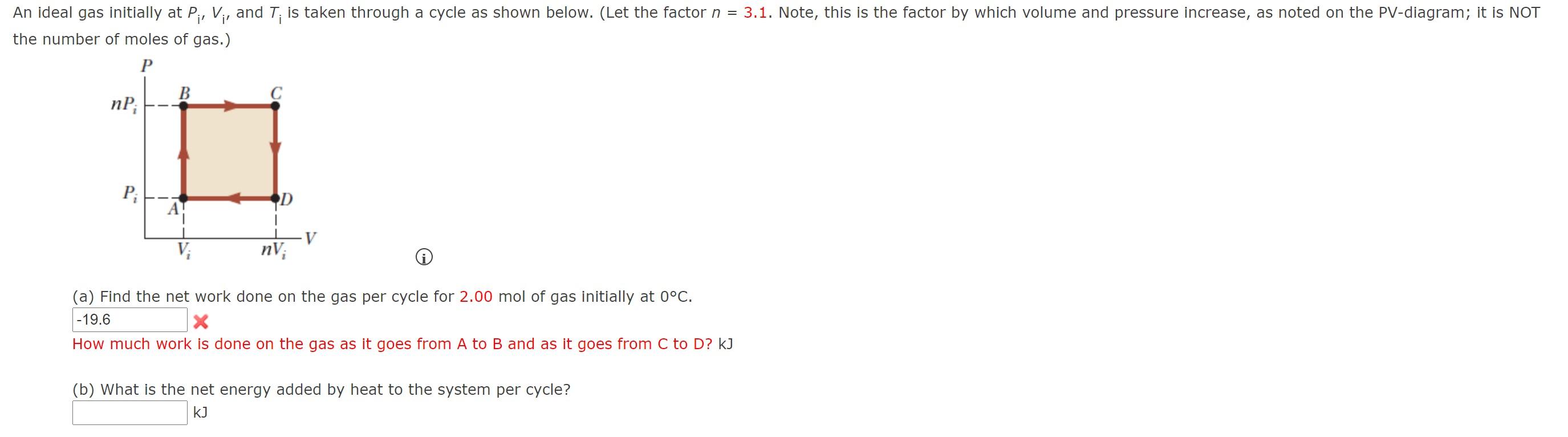
An ideal gas initially P_i ,V_i , and T_i is taken through a cycle
4.5 (412) · $ 8.00 · In stock
Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle

Solved An ideal gas initially at Pi, V;, and T; is taken

One mole of an ideal gas is contained in a cylinder with a movable piston. The initial pressure, volume,and temperature are P_i, V_i, and T_i, respectively. Find the work done on the

Solved An ideal gas initially at Pi, V;, and T; is taken

Chapter4, Lesson F - Performance of an Ideal Gas Cycle

Solved) - A 1.00-mol sample of a monatomic ideal gas is taken through the - (1 Answer)

Introduction to Thermodynamics

One mole of a gas in the state A(P1, V1, T1) is subjected to adiabatic

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done

An ideal gas expands isothermally from volume `V_(1)` to `V_(2)` and is then compressed to original
Solved An ideal gas initially at P_i, V_i, and T_i is taken
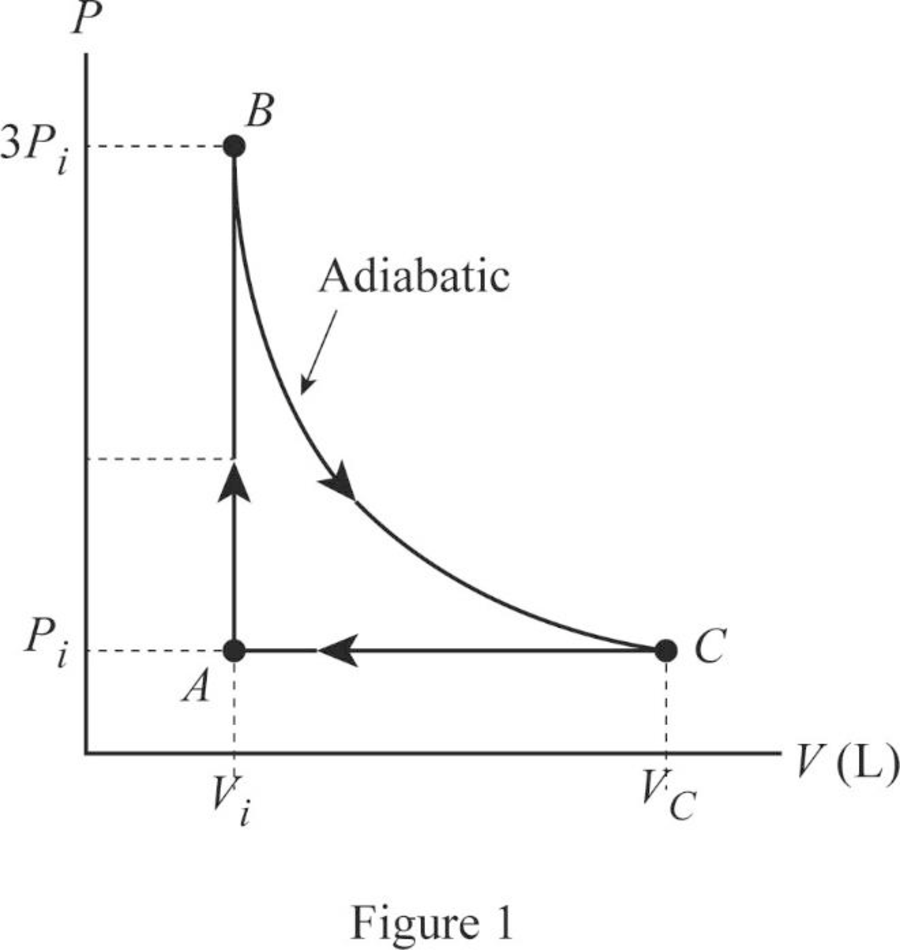
An ideal gas with specific heat ratio γ confined to a cylinder is put through a closed cycle. Initially, the gas is at P i , V i , and T i .

which is not really negligible with respect to unity Therefore the complete

P) Thermodynamics, PDF, Gases

solution manual for applied petroleum reservoir engineering by craft by kholoud hamad - Issuu
Isothermal Compression of a Ideal Gas and Distance