The correct order of increasing bond length of \( \mathrm{C
4.5 (223) · $ 15.00 · In stock
The correct order of increasing bond length of \( \mathrm{C}-\mathrm{H} \), \( \mathrm{C}-\mathrm{O}, \mathrm{C}-\mathrm{C} \) and \( \mathrm{C}=\mathrm{C} \

Solved QUESTION 4 Arrange the following bonds in order of

Forecasts for Galaxy Formation and Dark Matter Constraints from Dwarf Galaxy Surveys

SOLVED: Part A: Place the following in order of increasing bond length. C-C < C=C < C-N < C=O
OpenKIM · EAM Dynamo MendelevKramerOtt 2009 CuZr MO_600021860456_005 MO_600021860456 · Interatomic Potentials and Force Fields

BCF with bond length 0 = 0.48 (top) and 0 = 0.55 (bottom) at different
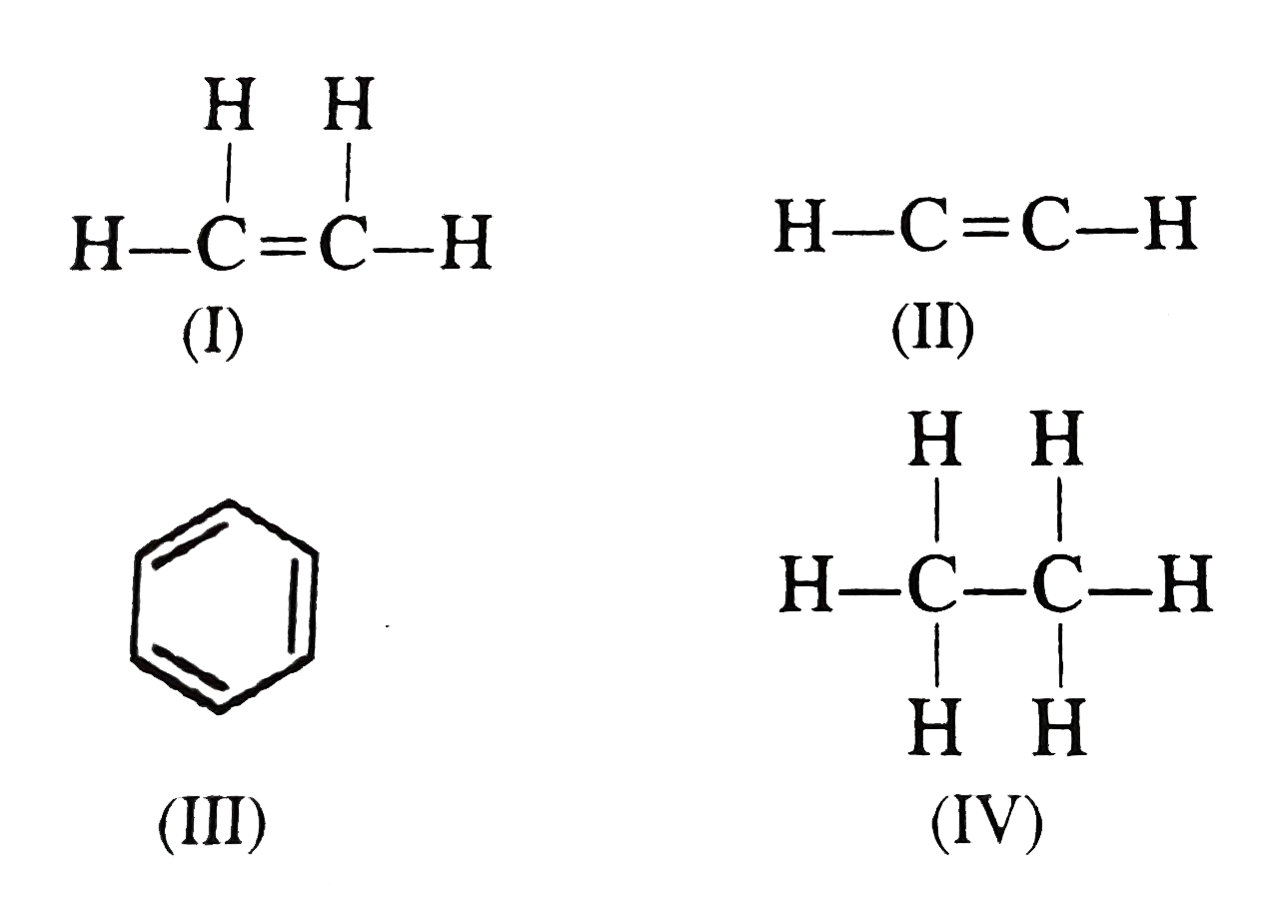
Decreasing order of C-C bond length is (I) C2H4 (II) C2H2 (III) C6 H6

The correct order of increasing bond length of C - H ,C - O, C - C and C = C is

Rank the following bonds in order of increasing bond length.

BONDING Let's get together… Barbara A. Gage PGCC CHM ppt video online download
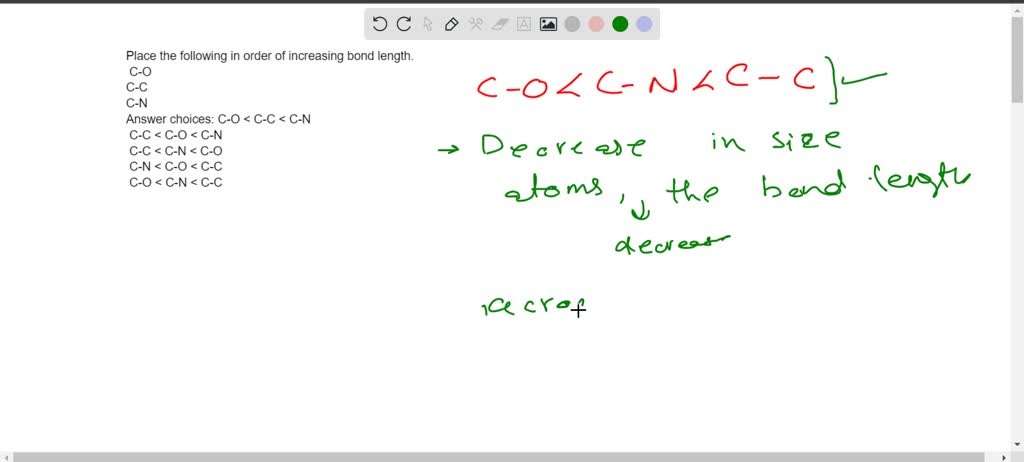
SOLVED: Place the following in order of increasing bond length. C-O C-C C-N Answer choices: C-O < C-C < C-N C-C < C-O < C-N C-C < C-N < C-O C-N < C-O < C-C C-O < C-N < C-C
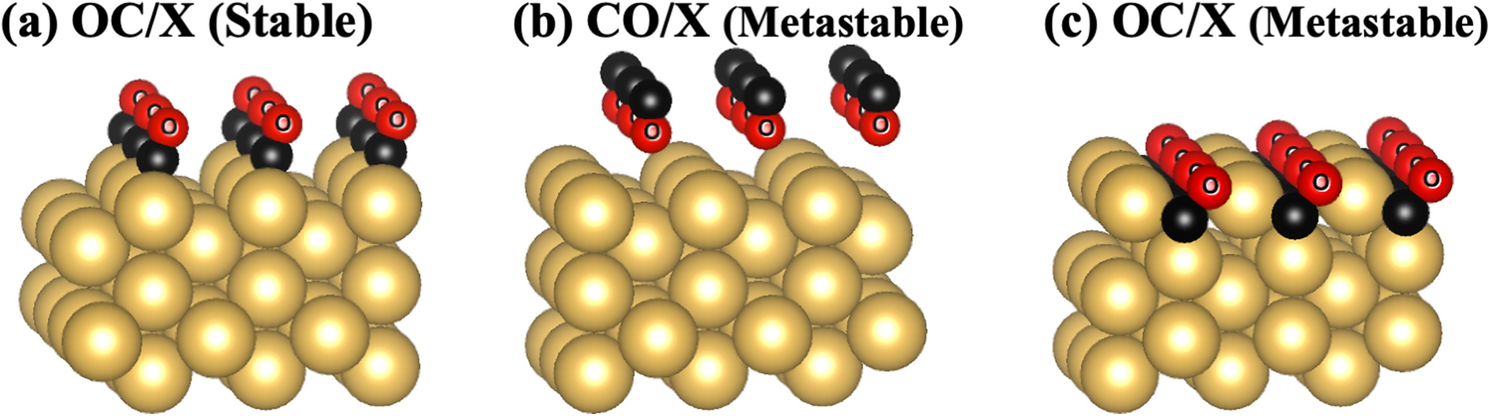
Carbon-oxygen surface formation enhances secondary electron yield in Cu, Ag and Au
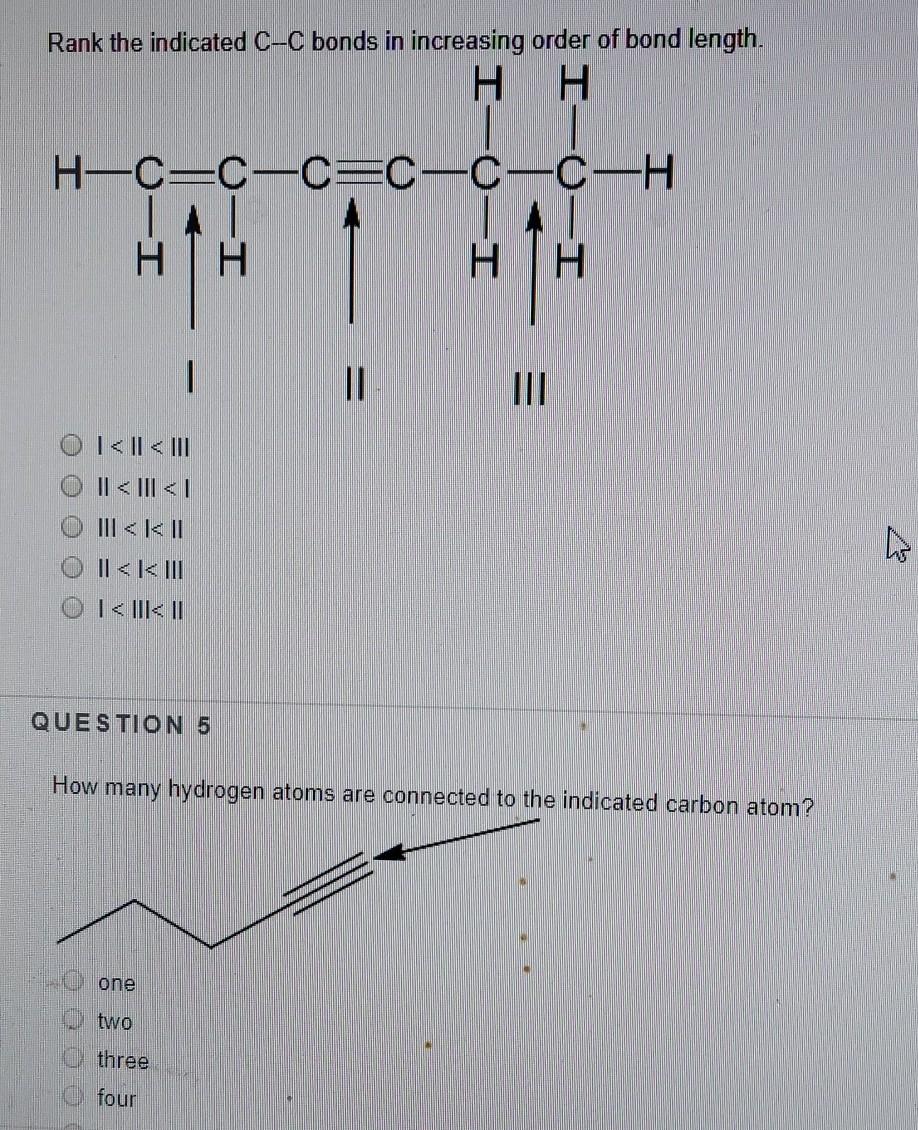
Solved Rank the indicated C-C bonds in increasing order of

Like several other bonds, carbon-oxygen bonds have lengths a












